K-Stem Cell, the first in the world to regenerate cartilages using stem cells
-
Presenting a fundamental treatment solution for degenerative arthritis through cartilage regeneration
A domestic research team has become the first in the world to develop a non-surgical method of treating degenerative arthritis by injecting stem cells into the joints.
The research team led by Dr. Jo Hyeon-cheol and Dr. Yun Gang-seop of the Orthopedic Surgery Science Department of the Seoul Metropolitan Government (SMG)-Seoul National University (SNU) Boramae Medical Center extracted mesenchymal stem cells from the abdominal area of degenerative arthritis patients and injected 100 million autologous mesenchymal stem cells into the knee joints after a cell culture process. They discovered that it led to the regeneration of cartilages above and below the knees with a volume increase of 14% and 22%, respectively.
High effectiveness achieved with an injection of at least 100 million stem cells
The research team conducted the study with 18 patients, who were divided into 3 groups: low dosage group (10 million stem cells), mid dosage group (50 million stem cells) and large dosage group (100 million stem cells). The results showed that cartilage regeneration and knee function improvement occurred most significantly in the large dosage group.
This was the world’s first clinical trial conducted in order to develop a stem cell “injection” for therapeutic purposes. There are currently no stem cell injections for knee arthritis treatment in the world. The conventional cell therapy “procedures,” which may seem similar to the therapeutic method developed in the aforementioned clinical trial, are a type of “medical practice” involving extraction of mononuclear cells from the bone marrow or adipose tissue to be injected at a later time. In this case, even if a million cells are injected, there are only 10 to 100 stem cells that actually serve useful. Plus, this is merely an estimation, and it is impossible to confirm how many stem cells were included in the injection. On the other hand, the stem cell “injection” used in this research is solely comprised of 100 million stem cells, which were strictly verified prior to the administration.
A single injection reduced the area of worn cartilages above and below the knees by 32% and 64%, respectively
The research team directly injected mesenchymal stem cells in the knees in which the bones were exposed due to worn-out cartilages. Then, 6 months later, the extent and quality of cartilage regeneration were observed through arthroscopy, MRI and histological examination.
The arthroscopic examination showed that the areas of born exposure above and below the knees decreased by 32% and 64%, respectively. On the other hand, the MRI exam showed an increase in the cartilage volumes above and below the knees by 14% and 22%, respectively, and the histological examination showed an increase in the volume of the femur cartilage by a whopping 300%. In addition, the results of a knee function test showed a 39% increase in the level of knee function and 45% reduction in the pain level. Based on these results, it was found that stem cell injections facilitate cartilage regeneration and restoration of knee functions.
In relation to the safety concerns, no adverse events were reported on any of the patients during the 12-month post-injection observation period.
A new era of stem cell therapy for degenerative arthritis
Degenerative arthritis is the most prevalent type of arthritis in the world. Degenerative arthritis affecting the knees is most common type of arthritis, occurring in about 37% of the population aged 60 and older. The fundamental cause of degenerative arthritis is the wearing of cartilages surrounding the joints. There have been no known drug therapies or surgical methods of regenerating cartilage in the form of synovial joint cartilage.
Thus, in the early stages of degenerative arthritis, the symptoms such as pain and edema are treated with analgesics and anti-inflammatory drugs combined with weight loss and physical therapy, while artificial joint replacement surgery is generally performed for severe cases.
This study is considered to have achieved a notable feat to the extent that it may completely shift the paradigm of degenerative arthritis treatment as it administered treatment using stem cell injections, which were considered only as possibility, to actual patients in a strictly controlled clinical trial, and scientifically proved the safety and effectiveness of the treatment, which gained international recognition.
The results of the research were published in “Stem Cells (Impact Factor 7.701),” an international scientific journal with authority in the field of stem cell research, in late January, and presented at Game Changers, a special session at the American Academy of Orthopaedic Surgeons Conference held in New Orleans in the U.S.A. in March 2014. Game Changers is a session for which the results of 15 studies, which can exert a substantial impact and shift the paradigm of musculoskeletal treatment within a few years, are chosen out of the 856 orally presented studies.
Dr. Yun, who headed the research project, said, “This study has significance as the world’s first commercial clinical trial related to the development of arthritis treatment method involving stem cell injections into the glenoid cavity. Above all, it is meaningful that the results of the study conducted in Korea were published in Stem Cells, an international scientific journal with authority in the field of stem cell research, and gained international recognition. Another significant factor is that the research was continued through a large-scale three-phase clinical trial to create a way to actually treat patients with knee arthritis in Korea with stem cells.” This showed that arthritis can now be treated through injection therapy, instead of surgical procedures.
Dr. Jo, who was personally involved in the research, stated, “There are no stem cell treatment agents, for which degenerative arthritis is the primary indication, that have received approval, anywhere in the world. This was the world’s most advanced research conducted on the development of a stem cell treatment method for degenerative arthritis so far”
Dr. Jo also added, “One of the biggest significances of this study is that it confirmed the three criteria for effective treatment of arthritis using stem cells. First, there is a need to thoroughly verify the stem cells being used are actually mesenchymal stem cells based on multiple scientific methods. Second, at least 100 million stem cells need to be injected. Third, an appropriate cell injection method and a post-treatment program must be administered.”
The SMG-SNU Boramae Medical Center is set to conduct a large-scale, three-phase clinical trial using autologous adipose-derived stem cells, in collaboration with prestigious university hospitals throughout the country, starting early this year. Patients wishing to participate in the clinical trial can receive stem cell therapy free-of-charge following a patient suitability assessment.
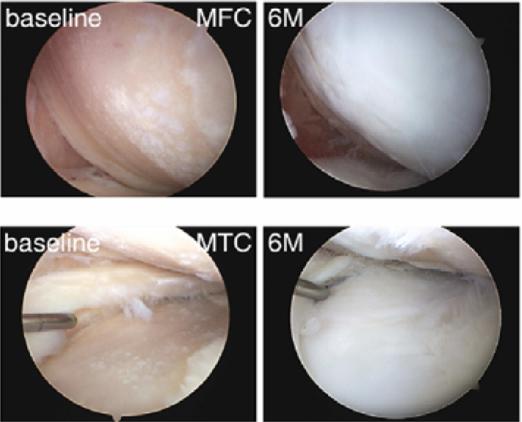
Photo description: The results of the arthroscopic observation showed regeneration of joint cartilage (white area) after 6 months.





대단한 연구성과입니다.
Reply우측하지 소아마비로 왼쪽 다리를 많이 사용을 하다 보니 왼쪽 무릎 연골이 많이 손상이 되어 있는 상태입니다. 혹 2차 임상 실험이 있으면 참가자로 지원을 해 보고 싶습니다.
Reply임상시험이 개시되면 홈페이지를 통해 환자 모집광고를 개제할 예정입니다.
Reply광고에 개재되어 있는 연락처를 통해 임상시험 참여가능 여부 확인이 가능하며, 임상시험 개시일에 대한 날짜는 홈페이지를 통해 추후 공지해 드릴 예정입니다.
감사합니다.